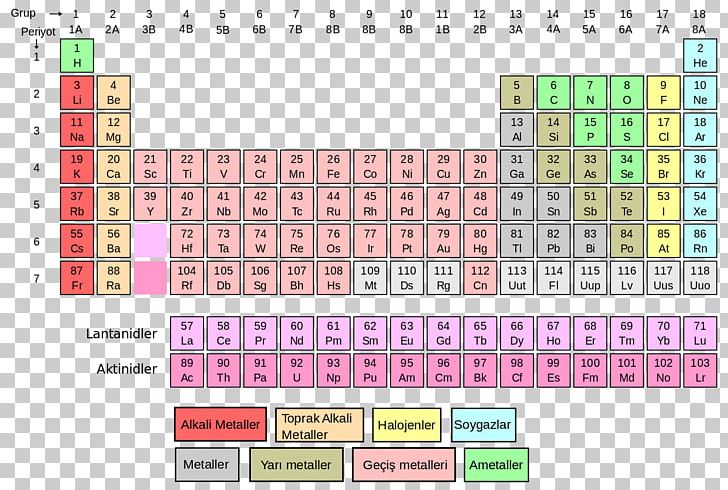 
is the process by which we learn about the natural universe by observing, testing, and then generating models that explain our observations. Science is the process by which we learn about the natural universe by observing, testing, and then generating models that explain our observations. This includes advances in medicine, communication, transportation, building infrastructure, food science and agriculture, and nearly every other technical field that you can imagine.Ĭhemistry is one branch of science. From the color that makes a rose so red to the gasoline that fills our cars and the silicon chips that power our computers and cell phones…Chemistry is everywhere! Understanding how chemical molecules form and interact to create complex structures enables us to harness the power of chemistry and use it, just like a toolbox, to create many of the modern advances that we see today. Section 2.1: Chemistry and Matter What is Chemistry?Įverything around us is made up of chemicals. Chapter 2: Atoms and the Periodic Table Section 2.1: Chemistry and Matter What is Chemistry? Physical and Chemical Properties Elements and Compounds Mixtures States of Matter Reactions in Chemistry Section 2.2: How Scientists Study Chemistry The Scientific Method 2.3 Atomic Theory with Historical Perspectives 2.4 Introduction to Elements and the Periodic Table 2.5 Dmitri Mendeleev and the development of the periodic table 2.6 Families of the Periodic Table 2.7 Defining the Atom Basic Atomic Structure – electrons, neutrons, and protons 2.8 Atomic Number – Protons Determine the Identity of an Element 2.9 Atomic Mass, Isotopes, and Molar Mass 2.10 Periodic Table Trends Atomic Size Electronegativity Ionization Energy Metallic and Nonmetallic Character 2.11 Chapter Summary and Homework 2.12 References
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
